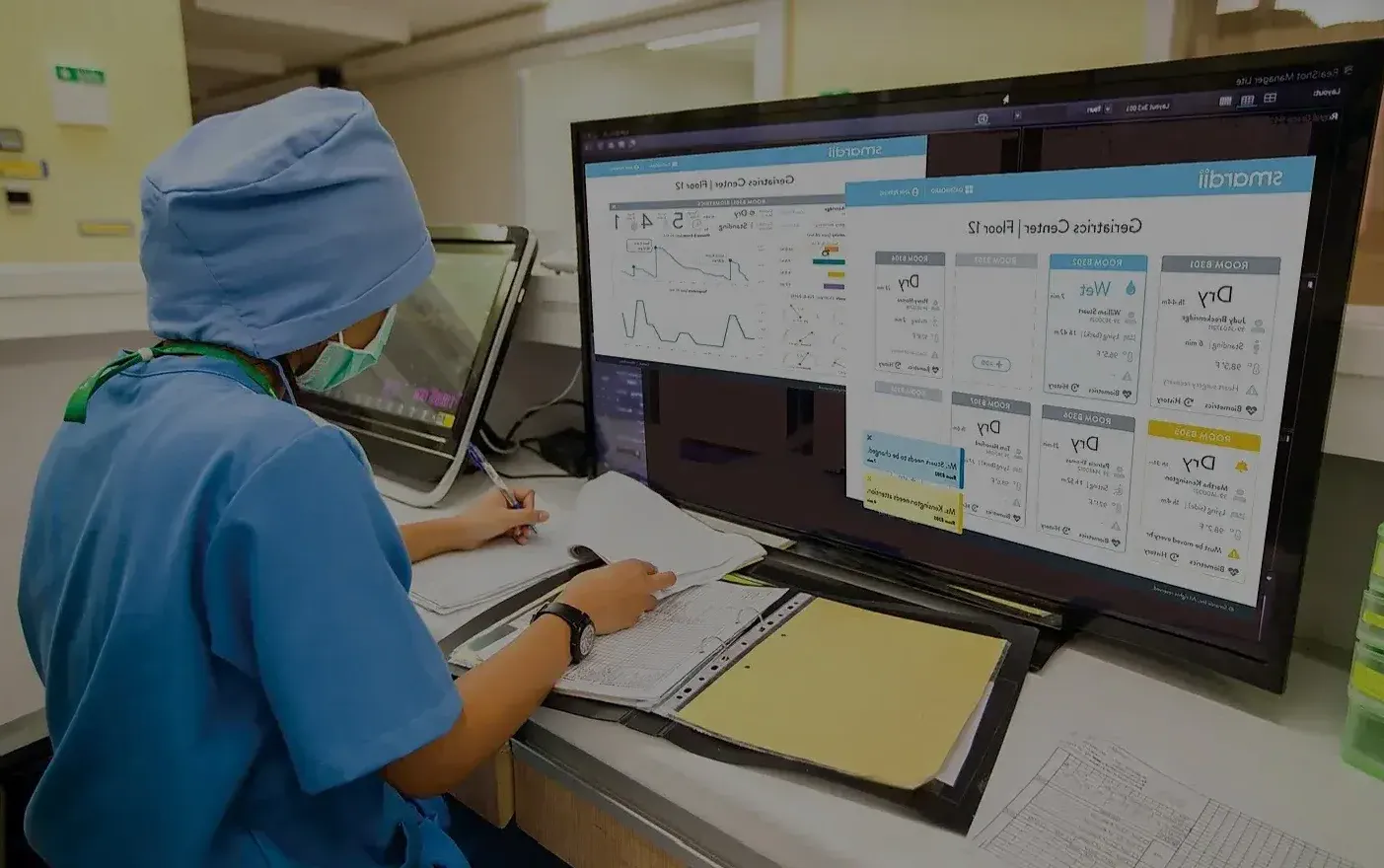
User Interface: What is it, and why is it important for medical device development?
In the rapidly evolving realm of medical device development, the concepts of User Interface (UI) and User Experience (UX) are indispensable, each carrying distinct significance. In this illuminating two-part series, we embark on an exploration to demystify these concepts and underscore their profound relevance to the world of medical devices, including the crucial integration of standards like IEC 62366.
Decoding UI Design: The Essential Connection between Clinicians and Devices
What exactly is UI design? In essence, the user interface serves as a vital conduit linking medical professionals and their devices, facilitating essential interaction. Consequently, UI design involves the intricate process of shaping the visual, tactile, and functional aspects of these interfaces. Within this realm, skilled UI designers take center stage, wielding substantial influence over the efficacy and user-friendliness of medical devices. The user interface of a medical device profoundly shapes clinical interactions, inherently affecting professionals’ efficiency and patient outcomes. With this in mind, designers endeavor to craft intuitive interfaces that offer seamless navigation, visually informative cues, and an overall gratifying user experience.
Exploring UI Design’s Clinical Dimensions
UI design delves into clinical intricacies, including:
- Thoughtful Color Schemes: Color choices go beyond aesthetics; they hold clinical significance. UI designers meticulously select colors that align with medical standards, ensuring that visual cues are easy to interpret. For instance, red might indicate alarms or critical notifications, while green signifies normal operating states. Compliance with standards like IEC 62366 ensures that color usage is consistent and meaningful.
- Purposeful Alarm Sounds: Alarm sounds are a crucial aspect of UI design, demanding careful consideration. These auditory cues must be distinct yet not jarring, promptly capturing clinicians’ attention without causing unnecessary anxiety. An intuitive distinction between urgent and non-urgent alarms can significantly enhance patient safety, a requirement emphasized by IEC 62366.
- Contextual Interaction: UI designers craft interfaces that acknowledge the clinical context. This means considering factors such as lighting conditions, the need for swift information retrieval, and minimizing cognitive load during critical procedures, aligning with IEC 62366’s focus on usability engineering.
- Workflow Integration: Medical devices are integral to clinical workflows. UI designers prioritize seamless integration, ensuring interfaces enhance, rather than disrupt, the natural rhythm of patient care, in compliance with IEC 62366 requirements.
- Patient-Centric Design: UI designers bear in mind that medical devices impact patients directly. Consequently, interfaces are designed with empathy, promoting patient comfort and engagement, as advocated by IEC 62366.
- Intuitive Navigation and Information Retrieval: UI design should prioritize intuitive navigation, allowing clinicians to swiftly access information they need. An organized layout, consistent icons, and clear labeling contribute to seamless interaction. User-centric design, wherein critical data is easily accessible during procedures, can significantly improve clinical decision-making, aligning with IEC 62366’s usability engineering principles.
Empowering Medical Devices with Expert UI Design
Elevating the performance of your medical device necessitates the expertise of adept UI designers. Accomplished designers understand the nuanced requirements of medical contexts, adeptly refining UIs to align with clinicians’ needs. With a legacy spanning over three decades, Nectar’s proficient team specializes in tailoring UI designs to the unique demands of medical devices. Witness the tangible impact of our expertise in this illustrative example. For a comprehensive exploration of UI design’s potential to optimize your medical device’s performance, we invite you to schedule a consultation here, ensuring compliance not only with IEC 62366 but also with industry best practices.
Incorporating these considerations ensures that UI designers for medical devices go beyond aesthetics, focusing on elements that enhance clinical functionality, user satisfaction, and patient outcomes, all while aligning with established standards like IEC 62366.
The Importance of Human Factors and IEC 62366
In the realm of medical device UI design, human factors take center stage. It’s not merely about aesthetics; it’s about ensuring that critical information can be swiftly and easily absorbed, even from across the room. Imagine a hospital setting where healthcare professionals need to monitor multiple patients simultaneously. The design of the user interface becomes a lifeline, allowing doctors and nurses to assess vital data with a mere glance. This aspect of UI design emphasizes the need for clarity, legibility, and intuitive layouts, ensuring that medical personnel can make split-second decisions based on the information presented, in line with IEC 62366’s human factors engineering guidelines.
It’s not just about making the UI visually appealing; it’s about saving lives through effective, user-centric design. IEC 62366 plays a pivotal role in achieving this by providing a framework for the application of human factors engineering to medical devices, ensuring that UI and UX design prioritize usability, safety, and overall user experience.
Navigating the Regulatory Landscape with IEC 62366
In the context of medical device development, adherence to regulatory standards is paramount. IEC 62366 serves as a cornerstone in this regard, offering guidance on applying usability engineering to medical devices. Compliance with such standards not only ensures the safety and efficacy of medical devices but also expedites the regulatory approval process.
- IEC 62366 Essentials: The IEC 62366 standard lays out essential principles for designing medical device user interfaces. It emphasizes the importance of conducting user research, risk management, and iterative design evaluations. This iterative approach aligns with the fundamental concept of continuous improvement, crucial for refining UI and UX.
- Enhancing User-Centered Design: IEC 62366 promotes a user-centered design approach. It encourages collaboration between engineers, designers, and healthcare professionals to create interfaces that address real-world clinical needs. This collaborative effort fosters innovation and ensures that the final product genuinely serves its intended users.
- Streamlining Regulatory Approval: Medical devices must undergo rigorous scrutiny before entering the market. Compliance with IEC 62366 helps streamline the regulatory approval process. Regulators recognize adherence to established usability engineering standards as a mark of commitment to safety and user experience.
- Patient Safety: Ultimately, IEC 62366 is about patient safety. The standard’s guidelines help prevent user errors, reduce the risk of adverse events, and enhance the overall quality of patient care. By aligning with IEC 62366, medical device developers prioritize the well-being of patients and healthcare providers.
The Nexus of UI, UX, and IEC 62366
As we delve deeper into the world of medical device development, it becomes evident that UI and UX design are intrinsically linked to the principles set forth by IEC 62366. These elements collectively contribute to the success of a medical device in the market and its impact on patient care.
- IEC 62366 as a Guiding Light: IEC 62366 serves as a guiding light for UI and UX designers. It provides a structured framework for incorporating human factors engineering into the design process. Designers can refer to the standard’s guidelines to ensure that their creations meet the highest standards of usability and safety, which are paramount in UX design.
- Iterative Improvement: Both UI and UX design are iterative processes. They involve continuous refinement based on user feedback and real-world usage scenarios. IEC 62366’s emphasis on iterative design aligns perfectly with the iterative nature of UI and UX design, fostering a culture of constant improvement and user-centered design.
- Human Factors Integration: The core of IEC 62366 is the integration of human factors engineering. UI and UX designers actively consider human capabilities and limitations, striving to create interfaces that accommodate users’ needs and capabilities. This human-centered approach is at the heart of both disciplines, ensuring that medical devices are not only functional but also user-friendly and safe.
Beyond Compliance: The Competitive Edge
While compliance with IEC 62366 is a regulatory requirement, it also provides a competitive edge in the medical device industry. A medical device with a user-friendly interface and a seamless user experience not only meets regulatory standards but also stands out in a crowded market. Healthcare professionals are more likely to embrace devices that enhance their workflow and improve patient outcomes.
In conclusion, the synergy between UI, UX, and IEC 62366 is undeniable in the field of medical device development. These elements, when integrated harmoniously, lead to the creation of medical devices that not only meet regulatory requirements but also exceed user expectations. In the dynamic landscape of healthcare technology, where innovation and patient safety converge, the importance of this synergy cannot be overstated. It ensures that medical devices not only save lives but also enhance the quality of care provided to patients worldwide.